H5N1 Analysis Request Form : https://www.hug.ch/laboratoire-virologie/centre-national-reference-pour-infections-virales
*Case definition for suspected viral respiratory disease: 1) ARI (inclusion criterion): acute respiratory disease defined as "a disease that begins acutely with cough, sore throat, shortness of breath or cold, and of infectious origin according to the physician's judgement‘. This definition is consistent with the WHO definition of ’Acute Respiratory Infections‘ (ARI). 2) ILI (optional criterion): ’ sudden onset of high fever (>38°C) with cough or sore throat".
For samples that are positive for influenza A and/or B, a detailed characterization of the influenza viruses present in the sample will be performed (subtyping, antigenic proximity to vaccine strains, genotyping and identification of mutations associated with antiviral resistance).
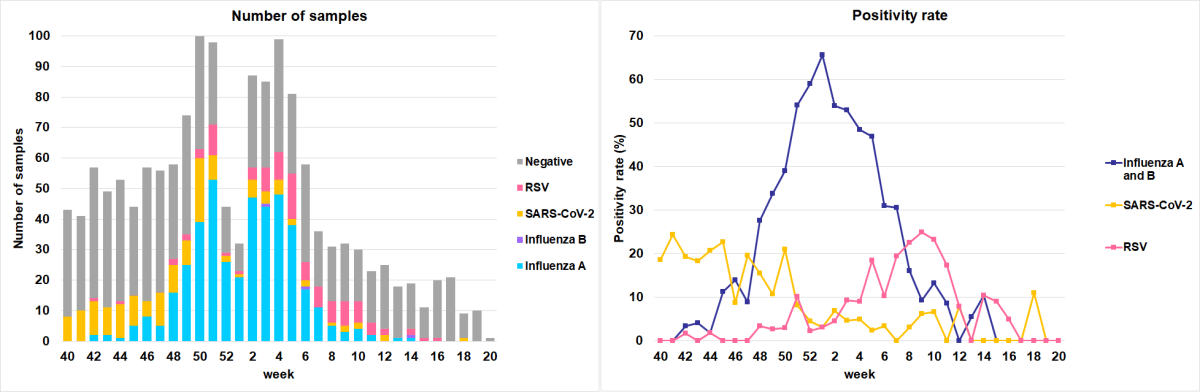
The surveillance season for respiratory viruses circulating among the Swiss population ended in calendar week 20 of 2026. Since calendar week 16 of 2026, the prevalence of respiratory viruses has been low. Updates will be provided monthly or in exceptional circumstances.
Note:
The data for the last two weeks are provisional.
The data are available on the FOPH dashboard for influenza and SARS-CoV-2.
The results of subtyping (A/H1N1 or A/H3N2) and lineage (B/Victoria or B/Yamagata) are available on the FOPH website here.
Contact :
