FAQs about the context of this clinical trial
- 1. Why did WHO ask the University Hospitals of Geneva to perform this clinical trial?
- 2. Why not test this vaccine in areas affected by the epidemic?
- 3. Will the future vaccines be given for free or will they be sold?
- 4. If WHO decides on a large scale vaccination campaign, how will priorities be set?
- 5. Why was this vaccine not been made available earlier to WHO?
- 6. Who is funding the clinical trial at the University Hospitals of Geneva?
- 7. Who funded the production of vaccines used at the University Hospitals of Geneva?
- 8. What other vaccines are being considered by WHO?
- 9. How can these vaccines contribute to stopping the current epidemic?
- 10. When and where will the other phase I clinical trials on the VSV vaccine take place?
FAQs about the clinical trial
- 11. How are the volunteers selected?
- 12. How often will the vaccinations take place?
- 13. When (and how) will we know if the VSV-ZEBOV vaccine works?
- 14. If the results are conclusive, will we need further clinical studies before implementing large-scale vaccination?
- 15. How much shorter is this process compared to the study of a conventional vaccine?
- 16. How many volunteers will receive a placebo?
- 17. Has anyone already received this vaccine?
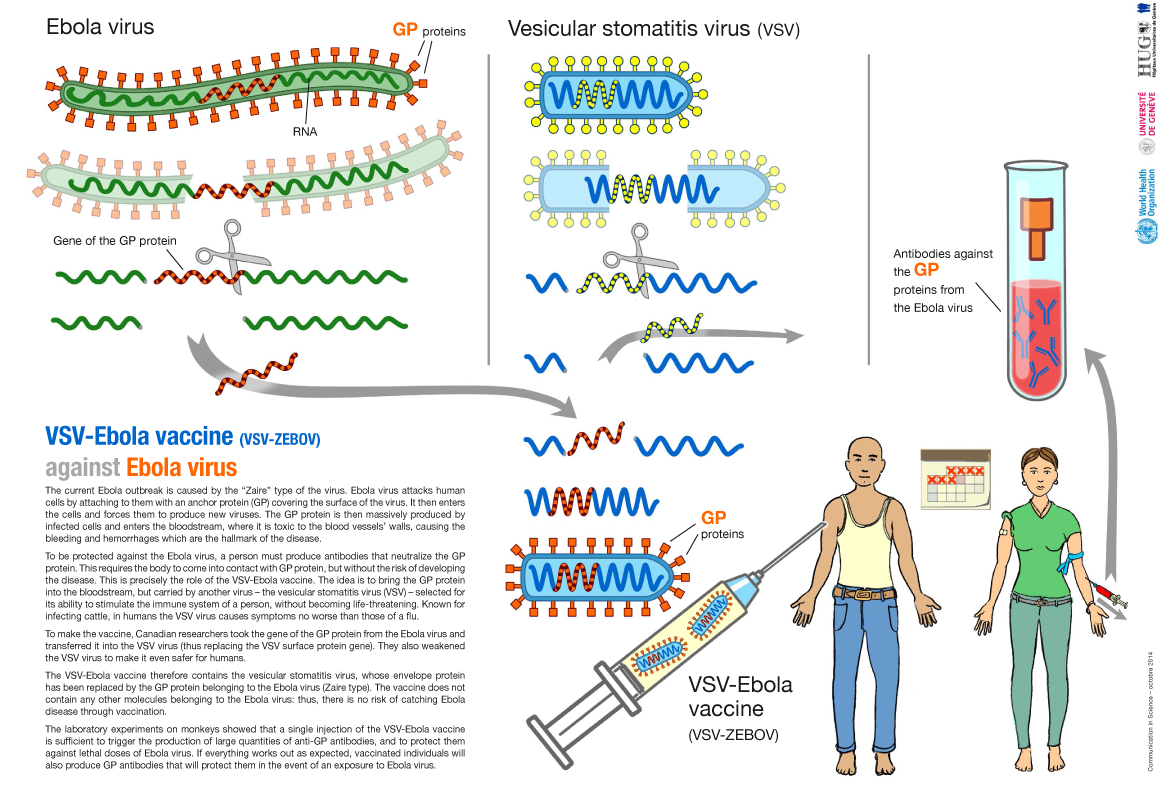
- 18. What do we know already about the VSV-Ebola vaccine?
- 19. Are the HUG in contact with the other teams that are testing this vaccine?
- 20. Is it be possible to modify the research protocol during the course of the clinical trial?
- 21. Have the HUG already performed this type of clinical trial?
FAQs on the VSV-ZEBOV vaccine against Ebola virus
- 22. How and by whom is this vaccine manufactured?
- 23. If the vaccine is "live", why is it not dangerous?
- 24. Has the vesicular stomatisis virus (VSV) already been used for other vaccines or therapeutic treatments?
- 25. Is this vaccine protected by "Intellectual Property Rights" which prevent other laboratories from producing it?
- 26. What are the odds for this vaccine being effective?
- 27. Could the Ebola virus mutate - like the flu virus - and therefore make the vaccine ineffective?
- 28. How long after vaccination would the protection become effective?
- 29. How long is the vaccine’s protective effect likely to last?
- 30. Will it be possible to vaccinate children? And what about animals, including those that could potentially transmit the virus to humans?
- 31. If the vaccine works well, is there hope that we could usefor people who have already contracted Ebola virus disease?
FAQs about the safety of the experimental vaccine
- 32. What side effects of VSV-ZEBOV vaccine candidate have been observed at the HUG?
- 33. What procedure was applied when these unexpected side effects were observed on some of the volunteers?
- 34. How do you know that these side effects were induced by the vaccination?
- 35. The vaccine is injected in the upper arm, so how is it possible to trace it in the joints or the skin?
- 36. Was the HUG team the only one to make such observations?
- 37. Was the clinical trial protocol modified on the basis of these observations?
- 38. Do we already know the results obtained with this lower dose of vaccine?
FAQs about the efficacy of the experimental vaccine
- 39. What was the intensity of the antibody response observed on volunteers?
- 40. Do we already know how long after the injection the person is potentially immunized, and for how long?
- 41. On the basis of these results, what dose of the vaccine has been selected for the next clinical trials in Africa?
- 42. Does this first clinical trial of a vaccine based on the vesicular stomatitis virus (VSV) open up other perspectives?