In Switzerland, epidemiological and virological surveillance of the influenza virus (flu) is provided by the Sentinella reporting system (1986) and the mandatory reporting system. Since 2018, a third system monitors influenza in hospitalized patients.
Of the primary care physicians (general practitioners, internists and pediatricians) participating in the Sentinella reporting system, around a half address nasopharyngeal swabs taken from some of their patients with influenza-like illness (ILI) to the National Reference Center of Influenza (NRCI). The latter performs the diagnosis for SARS-CoV-2, for influenza A and B and other respiratory viruses as respiratory syncytial virus (RSV), adenovirus, metapneumovirus, parainfluenza 1-4, rhinovirus and some respiratory enteroviruses, coronaviruses NL63/HKU1/229E/OC43 and bocavirus.
For samples that are positive for influenza A and/or B, a detailed characterization of the influenza viruses present in the sample will be performed (subtyping, antigenic proximity to vaccine strains, genotyping and identification of mutations associated with antiviral resistance).
On this page, you will find a report of the number of samples tested by the NRCI; as well as the results relative to viral characterization.
Report on the activity of influenza viruses (NRCI data)
Week 16/2024
(last update: 25.04.24)
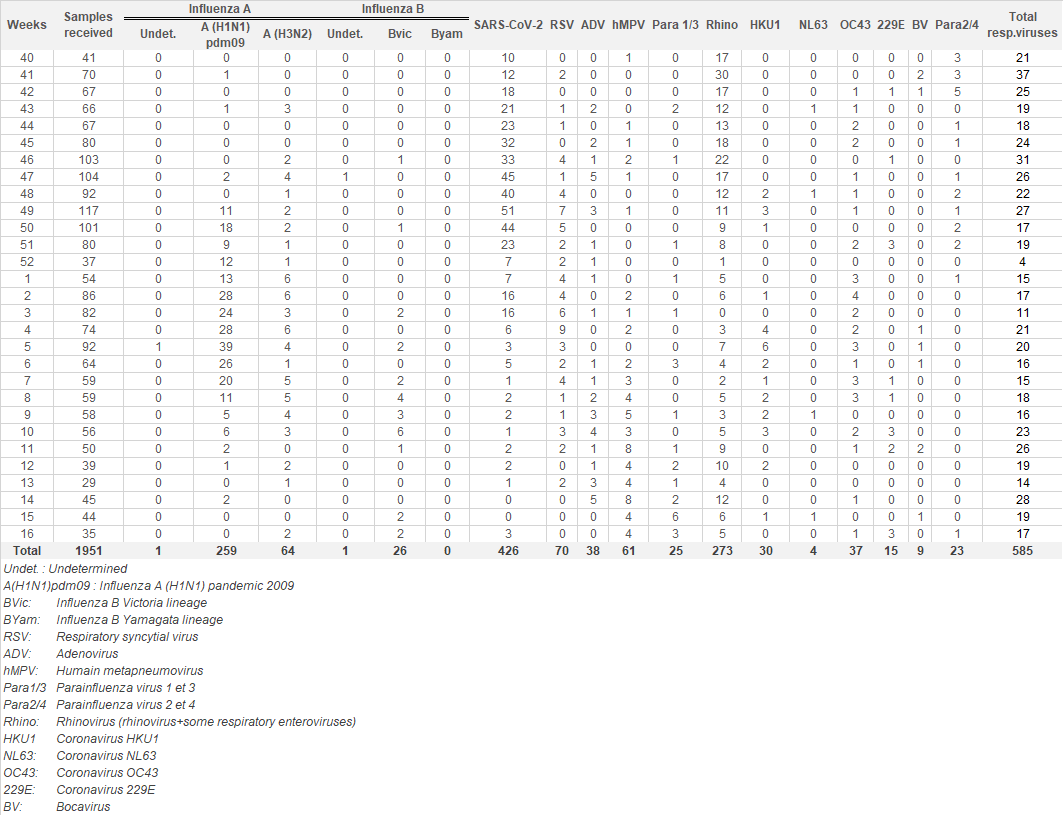
During week 16/2024, 35 samples from patients with suspected influenza or COVID-19 were analysed by the NRCI. Two influenza B viruses of Victoria lineage and 2 influenza A H3N2 viruses have been detected this week,representing a positivity rate of 11.4%.
Graph : Respiratory viruses detected from week 40/2023 to week 16/2024
Reduced influenza vaccine efficacy against influenza A(H3N2) viruses has been demonstrated during several consecutive seasons. This could be explained to some extent by:
Influenza A(H3N2) viruses are more variable than influenza A(H1N1)pdm09 and B viruses. Each modification/mutation in a virus is likely to cause differences between the vaccine strain and the influenza viruses circulating during the season. These are antigenic modifications; and for yet unknown reasons, they are more frequent in influenza A(H3N2) viruses than in the other two strains. From the time the influenza vaccine strain is recommended to the actual vaccination event (more than 6 months delay), A(H3N2) viruses are more likely to have undergone mutations, which may result in an antigenic drift potentialy impacting the vaccine effectiveness.
The main mode of production of vaccine strains remains their amplification in chicken eggs. But while all influenza viruses undergo mutations induced by this mode of production, changes in influenza A(H3N2) viruses have an increased tendency to result in antigenic modifications than those of A(H1N1)pdm09 and B viruses. Other production methods exist but are still not widely used.
Contact :
For further information on the activity of human influenza viruses in Switzerland and on the different tests and methods used to characterize them contact Ana Rita Gonçalves, 022 372 40 81 or
agnv@hug.ch .
Useful links (accessed 28.12.2023):


